|
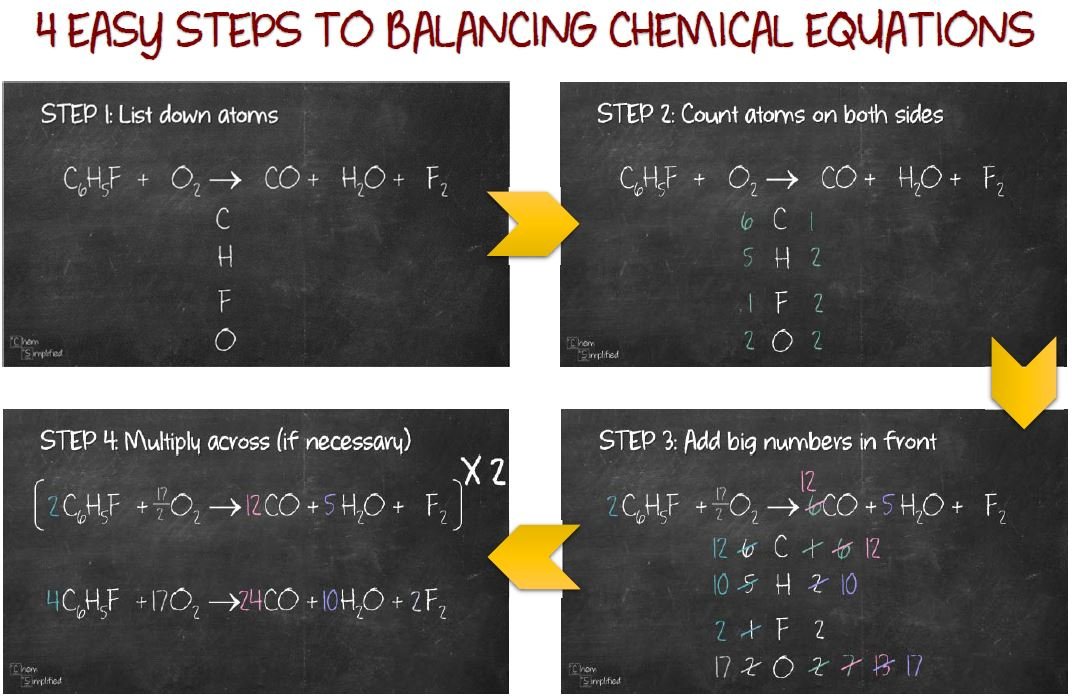
A video demonstration of the illustration can be found below. The illustration explores the process of balancing a chemical equation using the oxidation of a number of different hydrocarbons containing between one and five carbon atoms. The process of balancing an equation involves adding to each side of the equation until there are the same number of atoms of each element present on both sides. Without the proper number of reactants and products, a chemical equation is not a complete representation of the reaction. This is the significance of the numbers written before each compound in the reaction. A balanced chemical equation is one where there are the same number of atoms of each element on either side of the equation. To make sure that the equation conforms to the law of conservation of mass the equation must be balanced. A chemical reaction involves the rearrangement of atoms between molecules, not the creation or destruction of atoms. This mean that nothing can be gained or lost in the process. The law states that the mass of the reactants going into a reaction must be equal to the mass of the products. Properly written, the equation obey's the law of conservation of mass *. The chemical equation for the formation of sugar from water and carbon dioxide is:
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
